
Guest Lecture : Mechanism and Strategic Remedy
REPORT ON
Guest lecture
Topic: Echis Carinatus venom – induced sustained tissue decay: Mechanism and a strategic remedy
Preamble
This guest lecture was organized by the Undergraduate (UG) and Post-Graduate (PG) Departments of Biochemistry on 25th March 2026. The session aimed to bridge the gap between molecular biochemistry and clinical toxicology, focusing on the localized complications of snake envenomation.
Aim & Objectives
To identify why local tissue decay from Saw-scaled Viper (E. carinatus) bites is resistant to antivenom and to validate DNase I as a strategic remedy.
- IdentifyNETosis as the cause of vascular “clogging.”
- Contrast systemic vs. local toxicity (local being more dangerous for morbidity).
- Validate the “Rescue” hypothesis using a 10-day tail necrosis model.
Introduction
Snakebite envenomation is a critical public health challenge in India, primarily driven by the “Big Four” venomous species: the Indian Cobra (Najanaja), Common Krait (Bungaruscaeruleus), Russell’s Viper (Daboiarusselii), and the Saw-scaled Viper (Echis carinatus). While systemic complications like coagulopathy and neurotoxicity are the leading causes of death, sustained tissue decay (necrosis) at the bite site is the leading cause of permanent morbidity and amputation.
This report details the findings of a guest lecture regarding the unique pathophysiology of E. carinatus venom. It explores why conventional Polyvalent Antivenom Therapy fails to prevent local tissue destruction and highlights the role of the host’s immune response—specifically NETosis—as a key driver of this decay.
Details of guest lecture:
Date&Time:25.03.26,10:30am
Venue: MBA Conference Hall
Resourceperson:Dr. K. Kemparaju, Professor &Chairman , DOS in Biochemistry, University of Mysuru, Manasagangotri, Mysuru

The programme was inaugurated in the presence of Dr.NoorMubasheerCA,PGCoordinator and the resource person Dr. K. Kemparaju. Theprogram moderator, Ms. Harshitha Final year B.Sc Biochemistry student gave a brief introduction about the event. The invocation prayer song was sung by Alpha and team final year B.Sc Biochemistry students. The welcome address was given by,Mrs. Shruthi G, HOD, department of Biochemistry whowelcomed the dignitaries and the gathering of staff and students of Department of UG and PG Biochemistry, PG food science and nutrition and B.Voc Students.
The speaker for theguestlecturesession,Dr. K. Kemparaju, Professor &Chairman , DOS in Biochemistry, University of Mysuru, Manasagangotri, Mysuru,was introduced by Ms.SamyuktaMadhav Joshi,AssistantProfessor,Dept.of Biochemistry UG.
Dr. K. Kemparaju commenced the session by categorizing venomous snakes based on their toxin profiles. He provided a comprehensive overview of the “Big Four” in India but emphasized that snakebite is a global crisis.
To illustrate the severity of local toxicity, he presented two significant case studies from Africa. These cases highlighted a critical clinical failure: patients who survived the initial systemic hit (respiratory or circulatory failure) eventually suffered from horrific localized tissue decay. This established the premise that while we are good at keeping victims alive, we are currently failing to save their limbs.
The Clinical Conflict: Systemic vs. Local Toxicity
The lecture emphasized a major gap in current emergency medicine:
- Systemic Toxicity: Effectively neutralized by antivenom circulating in the blood.
- Local Toxicity: Often “antivenom-resistant.” By the time antivenom is administered, the venom toxins are already sequestered in the tissue.
- The Problem: Local toxicity is frequently more dangerous than systemic toxicity in the long term, as it leads to irreversible muscle loss, gangrene, and lifelong disability.
The Mechanism: NETosis and Venom Sequestration
The core discovery discussed was the role of Neutrophil Extracellular Traps (NETs).
- NETosis Induction: When the body detects E. carinatus venom, it triggers neutrophils to undergo NETosis. This process involves the release of DNA webs to “trap” the venom.
- The PMA Comparison: In a laboratory setting, PhorbolMyristate Acetate (PMA) is used as an industrial-strength inducer to study these traps. E. carinatus venom acts with similar potency.
- The “Clog” Effect: While NETs are meant to be protective, they actually block local blood vessels. This “clogs” the microcirculation, trapping the venom’s destructive proteases directly against the healthy tissue.
- Lack of DNase: Most snakes (Cobra, Russell’s Viper) have DNase activity in their venom that accidentally breaks down these traps. However, E. carinatus lacks this activity, meaning the DNA “webs” remain stable, keeping the venom trapped at the site and causing continuous decay.

Molecular Markers of NETosis
To quantify and confirm the presence of these traps, the research identified several key biochemical markers:
- Decondensed DNA Fibers: The structural “web” released into the extracellular space.
- Citrullinated Histone H3 (citH3): A hallmark of NETosis, where histones are modified to allow DNA decondensation.
- Neutrophil Elastase (NE) & Myeloperoxidase (MPO): Granule-derived enzymes that sit on the DNA fibers to kill pathogens (but here cause tissue damage).
- Peptidyl Arginine Deiminase 4 (PAD4): The essential enzyme responsible for histone citrullination and the initiation of NETosis.
Formal Treatment Hypothesis
The lecture proposed a logical “If/Then” framework for a strategic remedy:
The “Clog & Clear” Hypothesis
Pathology (The Problem):
E. carinatus Venom Massive NETosis DNA Webs Block Blood Vessels Sequestered Venom ToxinsConstant Tissue Destruction Tissue Decay (Necrosis).
Strategy (The Remedy):
Administer DNase I Degrades the DNA Backbone of the NETs No Blockage of Blood Vessels Restored Micro-circulation Venom is Flushed/Diluted No Tissue Decay.
The Challenge Study: The Tail Necrosis Model
To validate the hypothesis that NETs are the physical cause of tissue death, a 10-day longitudinal challenge study was conducted using a tail-injection model. This study compared
the physiological outcomes of envenomation over a 240-hour (10-day) period.
Experimental Groups & Observations:
| Group | Treatment | Observations (at 8 Hours) | Final Outcome (Day 10) |
| Control | PBS (Phosphate Buffered Saline) | No inflammation; normal blood flow. | Tail remained healthy and intact. |
| Venom Group | E. carinatus Venom | Rapid induction of stable NETs; vascular occlusion (clogging). | Total Necrosis: Sustained decay resulted in a detached tail portion. |
| Remedy Group | E. carinatus + DNase I | DNase I degraded the DNA webs; restored micro-circulation. | Recovery: The tail was rescued; tissue integrity was preserved. |
The study showed that within the first 8 hours, the venom triggers a massive “NETosis” event. Without intervention, these DNA traps act like a biological tourniquet. By Day 10, the lack of oxygen and the concentration of trapped toxins in the Venom Group lead to the physical dropping off (detachment) of the tail.
Conversely, in the DNase I Group, the “clog” was dissolved early enough to allow the body to flush out the toxins, proving that the decay is not inevitable if the DNA traps are removed.
Closing and Felicitation
Following a productive session, the formal closing of the event took place:
- Vote of Thanks:Mrs. Lydia Vandana delivered the vote of thanks, expressing gratitude to the speaker for his profound insights and to the management and faculty for their support in organizing the event.
- Felicitation: As a mark of deep appreciation, Dr. K. Kemparaju was formally felicitated by Dr. Noor Mubasheer CA (PG Coordinator) and Mrs. Shruthi G (HOD, Dept. of Biochemistry).

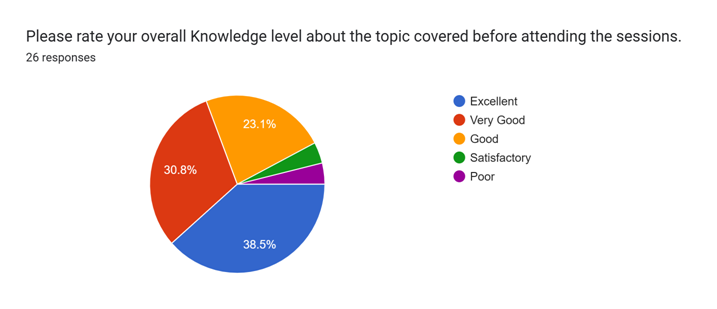
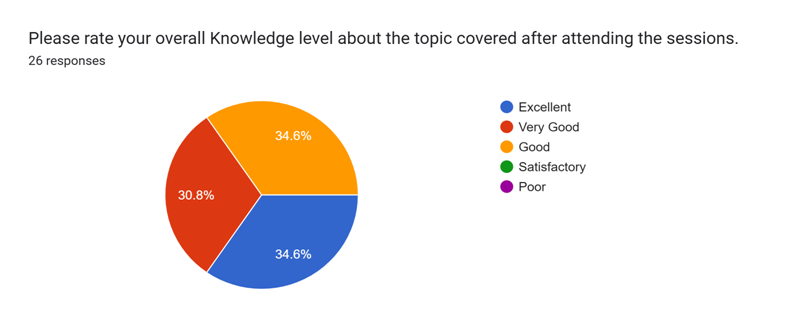
Feedback Summary
Conclusion
The lecture concluded that the tissue decay associated with Echis carinatus is not just a result of the venom’s potency, but a result of a “trapped” immune response. The stability of NETs, caused by the venom’s lack of DNase activity, creates a localized environment of death that antivenom cannot reach.
The Challenge Study provides a revolutionary strategic remedy: the use of DNase I as a co-therapy. By breaking down the physical DNA barriers (NETs), we can prevent the sequestration of toxins, thereby shifting the treatment focus from mere survival to the preservation of limbs and quality of life for snakebite victims.



